Understanding Clinical Trials in Lung Cancer
*Please note: This slide show is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always consult your doctor about any questions you may have regarding a medical condition.
What is a clinical trial for lung cancer?
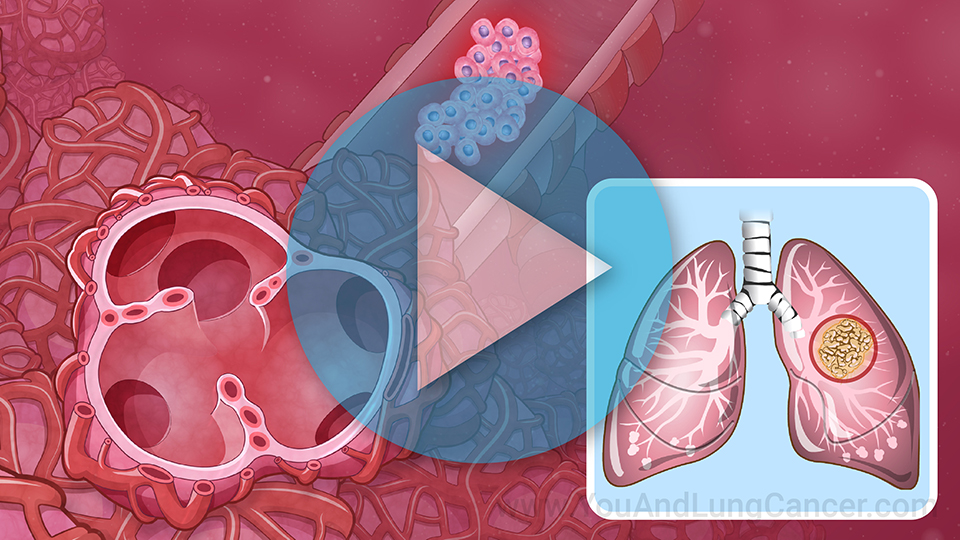
Lung cancer clinical trials are research studies in which people volunteer to help test new treatments, procedures, or ways to diagnose or screen for lung cancer.1
Why are clinical trials needed?
Clinical trials help researchers develop better tests and treatments.
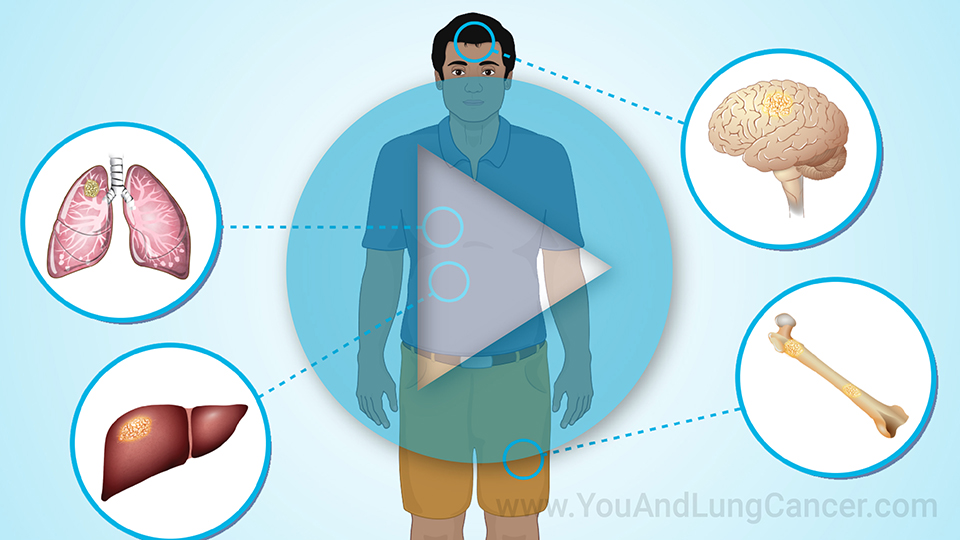
Clinical trials are especially important in lung cancer. They can help improve survival and quality of life for you and others with lung cancer.4
Trials also tell doctors about safety and side effects.2
What are pre-clinical studies?
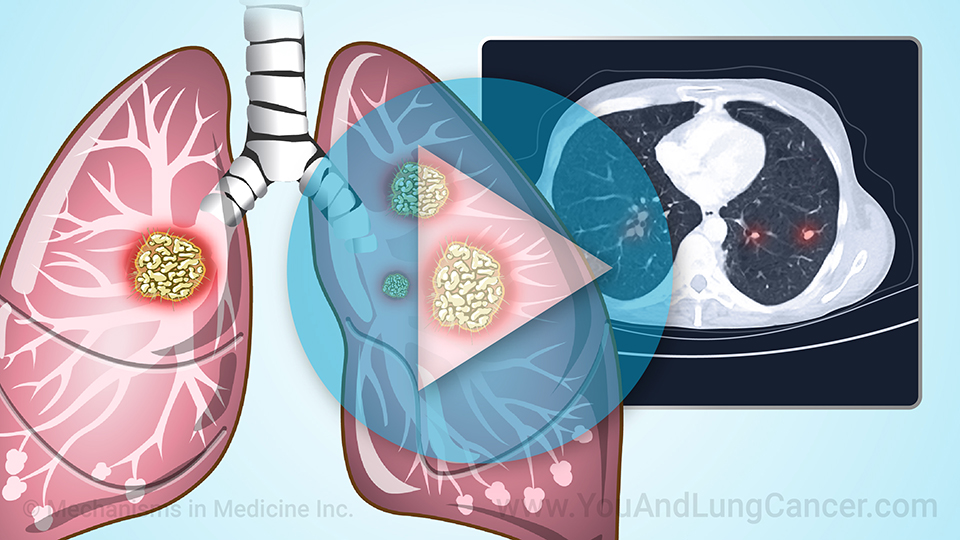
Clinical trial tests and treatments are studied before people volunteer to take part in the trial. "Pre-clinical studies" are done in the laboratory or on animals to demonstrate the rationale for testing in humans.
Both pre-clinical and clinical trials follow ethical and safety guidelines.
Who is in charge of clinical trials?
The Principal Investigator is the doctor responsible for the conduct of the trial.
Clinical trials are supervised in several ways. The research plan is reviewed by a group called an Institutional Review Board before the trial starts.
Experts called study monitors check during the trial to make sure the research is done correctly and that your information is kept private and secure. The trial can be stopped if problems are found.
Who approves treatments after clinical trials?
In the United States, clinical trial researchers ask a government agency called the US Food and Drug Administration (FDA) to approve the new treatment if it is found to be safe and effective. Then, doctors can prescribe it to people who need lung cancer treatment or screening.
Other countries have similar rules.
What are the phases of a clinical trial?
There are four phases of clinical trials. You may join a phase 1, 2, 3, or 4 clinical trial.
What is a Phase 1 clinical trial?
A Phase 1 clinical trial tests a new treatment in a small group of volunteers to learn about its safety and side effects.2
What is a Phase 2 clinical trial?
A Phase 2 clinical trial tests for effectiveness and side effects in a larger group of volunteers. It also gathers more information on safety.2
What is a Phase 3 clinical trial?
A Phase 3 clinical trial tests the treatment or procedure with hundreds of volunteers to confirm how well it works and compare it with available treatments. Researchers keep watching for safety and side effects.2
What is a Phase 4 clinical trial?
Phase 4 clinical trials are done after the new drug or treatment is approved. Researchers keep collecting information on safety, effectiveness, and side effects as the public begins using it.2
Who can be in a lung cancer clinical trial?
Each clinical trial has rules about who can participate. In research, these rules are called eligibility criteria or inclusion criteria. Researchers need different people to take part in different clinical trials. For example, a particular trial may need volunteers with a certain type or stage of lung cancer, or of a certain age, sex, or ethnicity.
Each trial also has specific exclusion criteria – which are circumstances that would prevent patients from participating in the trial.
How can I join a lung cancer clinical trial?
Ask your doctor about joining a clinical trial. You can ask anytime before or during treatment.
You can also search online for clinical trials in lung cancer. In the United States, ClinicalTrials.gov is a reliable place to look.
You can also contact A Breath of Hope Lung Foundation at abreathofhope.org/clinical-trials or sign up at ResearchMatch.org.
What is informed consent?
You must learn about a trial before deciding to take part. You talk with a study coordinator or doctor, review the informed consent form, ask questions, and only sign an agreement if you decide to join the trial.
The process of learning about the trial and then signing an agreement is called informed consent. In order to participate in a clinical trial, you must sign the informed consent form.
Can I leave a clinical trial after I join?
You can leave a clinical trial at any time.7 You will still receive treatment for your lung cancer unless you want to stop that too. Talking with your doctor can help you make these decisions.
What is a placebo?
A placebo is something that looks like the treatment being tested but is made of inactive ingredients.8 Using a placebo is often the best way to know if the addition of a drug is truly effective.
It is considered unethical to give a patient a placebo as the only therapy on a trial if there are already approved standard treatment options for a patient. Placebo trials may be used if the patients getting the placebo are receiving an approved standard treatment or if there are no available standard treatments.
Benefits and risks of taking part in a clinical trial
The benefit of taking part in a clinical trial is that it allows you to try a drug, treatment, or procedure that is not yet available to the public. The risk is that you may experience unwanted side effects.7
The trial may benefit your health, or it may not. But the knowledge that researchers gain will benefit people in the future.7
References
- Medline Plus. Clinical trials. Last updated August 31, 2021. Available at: https://medlineplus.gov/clinicaltrials.html. Accessed February 7, 2022.
- U.S. National Institutes of Health. The Basics. NIH Clinical Research Trials and You. Last reviewed November 10, 2021. Available at: https://www.nih.gov/health-information/nih-clinical-research-trials-you/basics. Accessed February 7, 2022.
- Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65(2):87-108. doi:10.3322/caac.21262
- U.S. National Institutes of Health National Cancer Institute. Advances in lung cancer research. Updated October 26, 2021. Available at: https://www.cancer.gov/types/lung/research. Accessed February 7, 2022.
- Dana-Farber Cancer Institute. How do I know if I am eligible for a clinical trial? Insight blog, May 10, 2019. Available at: https://blog.dana-farber.org/insight/2019/05/how-do-i-know-if-i-am-eligible-for-a-clinical-trial/. Accessed February 8, 2022.
- UpToDate. Patient education: What are clinical trials? The basics. Available at: https://www.uptodate.com/contents/what-are-clinical-trials-the-basics. Accessed February 7, 2022.
- ClinicalTrials.gov. Learn about studies. Available at: https://beta.clinicaltrials.gov/about-studies. Accessed February 7, 2022.
- U.S. National Institutes of Health. National Cancer Institute. Use of placebos. Reviewed February 6, 2020. Available at: https://www.cancer.gov/about-cancer/treatment/clinical-trials/what-are-trials/placebo. Accessed February 8, 2022.
This slide show explains what
clinical trials are, how they are conducted, and why they are important for patients with
lung cancer.
Lung cancer clinical trials are research studies in which people volunteer to help test
new treatments,
procedures, or ways to
diagnose or
screen for lung cancer.
Clinical trials for lung cancer are needed to
find new lung cancer treatments,
safer treatments, and more. This slide show also provides an overview of
study design,
eligibility criteria,
informed consent,
safeguards, the
phases of clinical trials, and the potential
benefits and
risks of participation. Be sure to ask your doctor about joining a clinical trial for your lung cancer. You can ask anytime before or during treatment.
-
Share with family and friends:
Click here to take our SURVEY
Your feedback is important to us! We will use your feedback to develop future areas of content about lung cancer which will help other patients, caregivers and families.